Exploring the link between our guts and our general health is becoming increasingly popular. Studies of people with various physical and mental health conditions suggest there may be an important link that has not yet been explored in MND.
Researchers are now looking closely into the association between our gut microbiome and our vulnerability to develop a range of psychological and neurological conditions, ranging from autism, depression, schizophrenia, to multiple sclerosis, Parkinson’s disease and MND.
Now more than ever, research into finding out more about the impact of our microbiome on our mental and physical wellbeing is being carried out, with more than 80% of all scientific publications on gut microbiome being published after 2013. This surge of interest in the topic is quite optimistic and has the potential to repair any functions affected by the ill-effects of gut imbalance.
The good bacteria
The gut microbiome is a collection of microorganisms, such as bacteria, fungi or viruses that live in a symbiotic relationship with its host – the gut. Although often overlooked, this system houses as many microbial ‘hitchhikers’ as we have cells in our body, and it collectively weighs more than the average human brain (around 2kg/4.5 lbs).
 Contrary to what some might believe, these microorganisms, or microbiota, don’t necessarily cause a riot in our body, but are rather important for our survival, helping with the breakdown of certain substances, protecting us from harmful organisms and providing protection to our cells.
Contrary to what some might believe, these microorganisms, or microbiota, don’t necessarily cause a riot in our body, but are rather important for our survival, helping with the breakdown of certain substances, protecting us from harmful organisms and providing protection to our cells.
Our microbiome starts being shaped from the moment we are born, factoring in the method of childbirth, microorganisms in our mother’s breast milk, and our surroundings. Although it is thought to broadly stabilise by the age of three, the journey doesn’t end there and our environment and lifestyle throughout our lives, such as exposure to a specific diet or medications, also play an important, although steadier, part.
This great number of factors therefore makes our microbiome unique from any other person, something which might explain some of our intolerances and dislikes. Be it in our gut, skin or mouth, microbiota affect the way our body deals with most of the things we are exposed to.
Is my gut talking to my brain?
Although connection between the gut and a number of health conditions has been suggested, the precise mechanisms aren’t yet clear. But perhaps the question most people might ask is ‘how can the gut affect anything so seemingly distant and unconnected as the brain?’
This happens through a system called the gut-brain axis, which allows communication between the neurones in the brain and spinal cord (the central nervous system) and the gut (the enteric nervous system) via multiple channels that regulate important functions such as hormones, immunity, digestion, or metabolism.
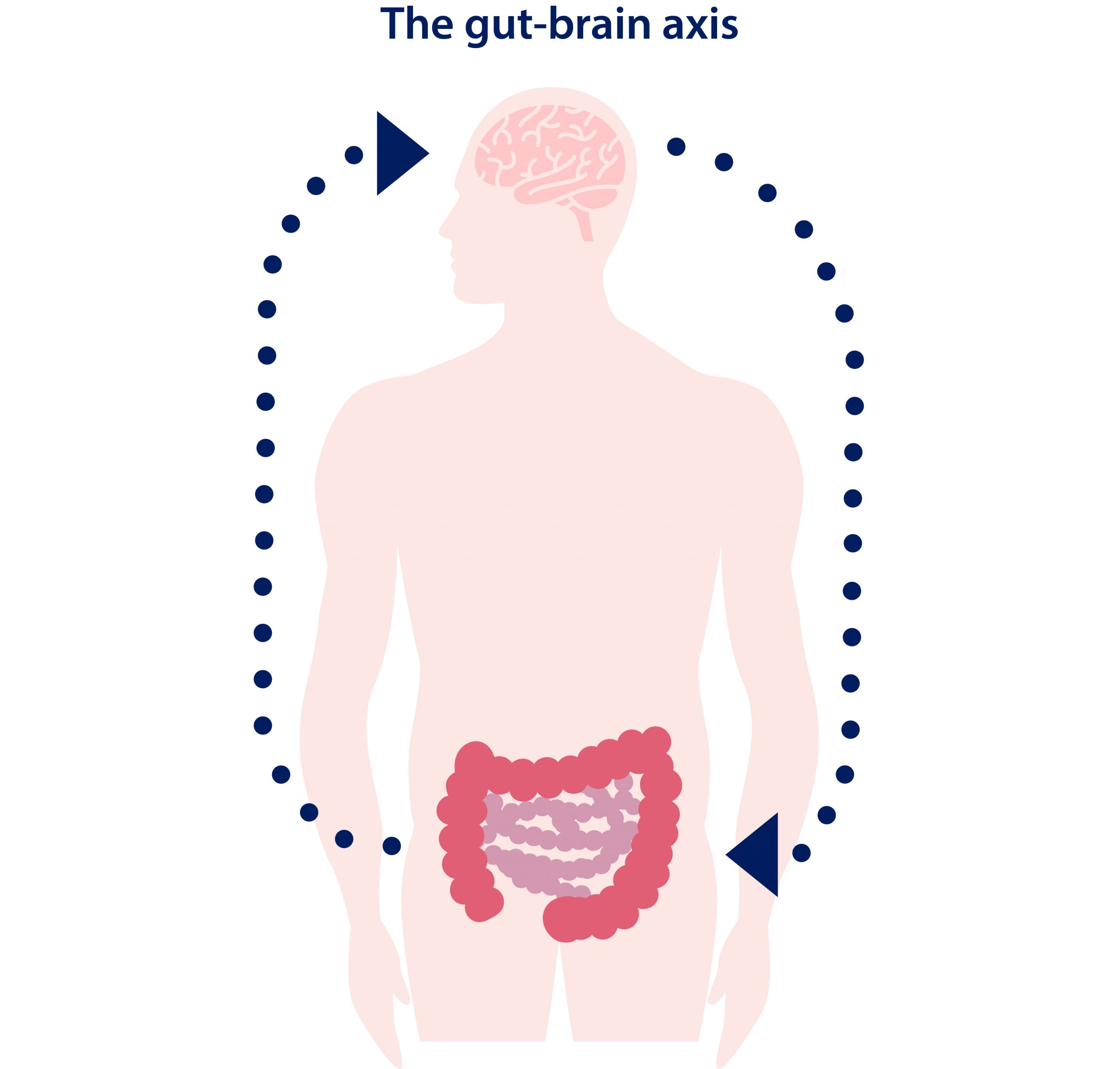
In order for this system to work correctly, the gut microbiome needs to be well balanced. When this balance is impaired, be it due to environmental, lifestyle or genetic influences, the gut finds itself in dysbiosis, a state of imbalance in which some of the microbiota are impaired and not working properly. When the microbiota try to influence the local environment of the gut through affecting its own (enteric) nervous system, the bacteria may inadvertently have a knock-on effect on the central nervous system. A consequent cascade of actions may then contribute to conditions of neurological origin.
MND and the gut
To date, most reports of gut imbalance in people with MND are based on small, sometimes contradictory studies, and investigations into whether there is any direct (or indirect) association are currently being conducted. However, when looking at rodent models, some studies have found a change in the diversity of the microbiome of MND mice compared to those without the disease. Specifically, reduced amounts of butyrate-producing bacteria and increased intestinal permeability (also known as ‘leaky gut syndrome’) have been detected, which was observed even before the onset of symptoms. (Although similar findings were found in a small study with people with MND, more studies need to be done to confirm this.)
Butyrate is mostly received from food or supplements, but our microbiome also has the ability to create it from fibre. It is therefore possible that when specific bacteria that help to produce butyrate are reduced in the microbiome, it can lead to problems with digestion, reduced anti-inflammatory abilities or lack of regulatory T-cell (Treg) production.
The important role of Tregs in MND has already been suggested. Researchers believe the gut bacteria influence the brain by using the immune system (Treg cells specifically) in the blood as an intermediary. High Treg levels in the blood have been linked with slower disease progression in MND and researchers think this occurs by somehow reducing the amount of inflammation happening in the brain and spinal cord. Some clinical trials (such as MIROCALS) are already investigating whether increasing the number of Tregs can reduce neuroinflammation.
Lack of butyrate is however only one of the possible affected pathways and the microbiome is now being closely investigated for any unique changes that only occur in people with MND. In the UK, for example, Dr Nik Sharma and his team are currently looking into the composition of the gut microbiome of people with MND and how this can be utilised to search for treatment (find out more about the study).
To explore this topic further and to find out more about the impact of the gut on our health, watch Dr John F Cryan of University College Cork (Ireland) talking about the way microbes can change our mind.
Publications and resources:
- Cani, P. (2018) Human gut microbiome: hopes, threats and promises. Gut
- Marietta, E., Horwath, I and Taneja V. (2018) Microbiome, immunomodulation and the neuronal system. Neurotherapeutics
- Sarkar A. et al. (2016) Psychobiotics and the manipulation of bacteria-gut-brain signals. Trends in Neuroscience
- Rea, K., Dinan, T. and Cryan, J.F. (2016). The microbiome: A key regulator of stress and neuroinflammation. Neurobiology of stress
- Hardvard Medical School: The Gut and the Brain





Thank you Craig, it is a truly fascinating area of research and we are hoping to know more about this connection in the near future. Hopefully, we will be able to follow up with blog reports from any ongoing microbiome studies.
It is good to see that your own personal ‘experiment’ has made a difference to you – after all, all of our bodies are unique and we will all react slightly differently to different foods.
We will keep a close eye on the topic and report back any updates!
Sir i from pakistan my brother is suffering from MND .can u people delivered me a treatment as pakistani doctors have failed.
Dear Shoaib,
Please can you email us at research@mndassociation.org so that we can provide you with answers and more information?
Thank you,
Martina
Very interesting post, thank you. I’m currently looking closely at my food intake to see if there is any positive or negative impacts on my MND. Nothing else has helped so far, so my daily food intake is the next course of action.
Research Team, this bit very interesting. I was formally diagnosed with MND 5yrs almost today I had feeding peg 2yrs ago but not using it yet. I eat everything if mashed up.
I do have stomach issues as can be constipated for up to three days. I record everything on my calendar in old phone and as of today not been for three days. After first day I take 10ml Senna and second time Senna tablet and third both and that works usually.
I drink at least 3ltr of water a day and still bladder works well but sometimes in night while asleep do have accidentally pee.