Dr Arpan Mehta, one of our Lady Edith Wolfson Clinical Fellows, and his team at the Euan MacDonald Centre at the University of Edinburgh have recently carried out a systematic review and meta-analysis of the pre-clinical literature (studies using animal models) to assess the therapeutic potential of targeting mitochondrial dysfunction in MND, examining if these interventions significantly affect survival in animal models of the disease, and determining the most effective time to begin treatment.
Why is this important?
In-depth, structured and unbiased analysis of pre-clinical studies that have already been completed is essential to advancing and accelerating drug discovery. These enable researchers to ‘ask the right questions’ both pre-clinically and when developing potential new treatments to take into clinical trials. By carefully analysing other studies at the pre-clinical stage, and by finding what works, they can use this information to identify the best potential therapies that may yield better results at the clinical trial stage.
Dr Arpan Mehta says: ‘This is the first comprehensive review and quality assessment of the pre-clinical ALS literature examining the influence of interventions targeting mitochondrial dysfunction on survival. We hope that it stimulates researchers to focus more on studying mitochondrial dysfunction in ALS/MND. Our findings have also allowed us to make recommendations guiding how future research should be conducted to improve the quality of the conclusions that can be made. Ultimately, this should increase the chance of translating successful findings in the laboratory to effective therapies in the clinic’.

A little bit of science to get us started …
People with MND often experience weight loss, hypermetabolism and high cholesterol. Metabolism is the term used to describe all the chemical reactions involved in maintaining the living state of the cells and the organism. Hypermetabolism is an increased rate in metabolic activity leading to an increase in basal metabolic rate – the total number of calories needed to perform basic, life-sustaining functions. Simply put, the body uses more calories for energy than the person takes in.
Mitochondria are specialised structures within a living cell that are essential for the regulation of metabolic pathways and cell survival. Processes called the Krebs Cycle and oxidative phosphorylation convert nutrients from food to ATP, a chemical that provides energy to fuel essential cellular activities, like sending messages that make muscles move. Mitochondria also play a central role in maintaining the correct balance of calcium in the body (calcium can lower cholesterol levels) and they regulate normal cell death.
In people with MND, the cellular distribution of mitochondria is affected and the total amount of mitochondrial DNA (mtDNA) is reduced in the spinal cord of people with the disease. mtDNA play a role in oxidative phosphorylation so defects in mtDNA may result in decreased activity, particularly in tissues with high energy expenditure such as those of the brain and muscle.
What did the team do?
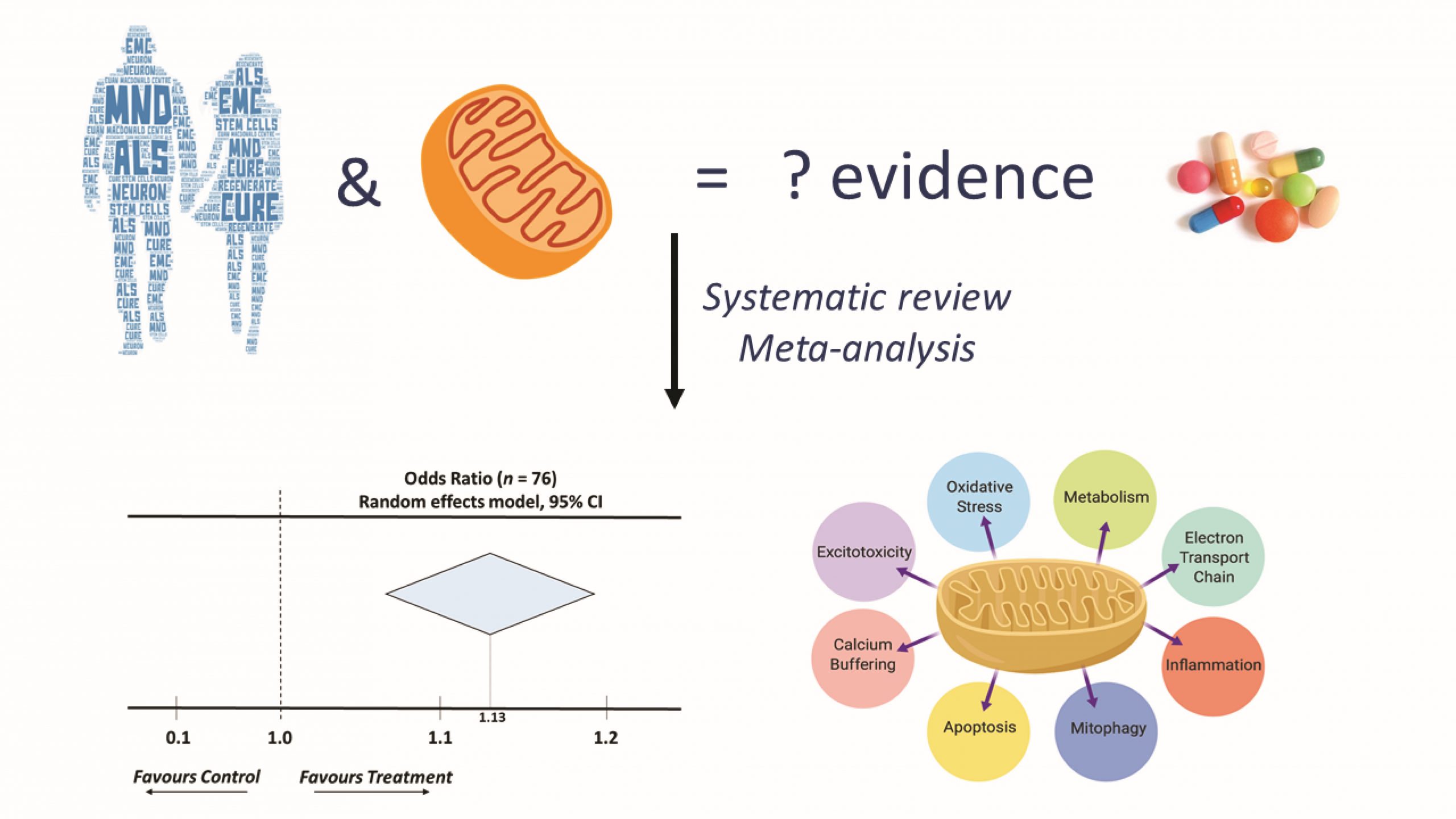
They identified 76 papers that met their inclusion and exclusion criteria and used meta-analysis (the examination of data from a number of independent studies of the same subject, in order to determine overall trends) to establish if interventions targeting mitochondria were effective and if the timing of the intervention – pre-, at- or after-symptom onset – influenced the effectiveness of the treatment.
What did they find?
Meta-analysis of MND animal studies conducted showed that drugs targeting mitochondrial dysfunction result in a statistically significant improvement in the survival of the animals. The greatest benefit was seen when the therapy was delivered prior to onset of symptoms. When interventions were delivered at later time points, this statistically significant effect went away, but this is likely to be because of the relatively small number of studies available that looked at these later time points. It was therefore not possible for the team to make a definitive conclusion about how effective therapies were that were administered later.
By further analysing different target pathways and whether interventions were directly targeting mitochondria (only 7 studies), or indirectly through cell-wide distribution (69 studies), they found that three quarters of the studies with a cell-wide distribution demonstrated effectiveness, as did all of the mitochondria-targeting interventions.
What does this mean for future MND research?
Dr Mehta and his team have demonstrated that there is likely to be potential in targeting mitochondrial dysfunction in MND, particularly if the treatment is delivered early. These findings offer promise both in terms of forming a basis for future MND therapies and encouraging research that focuses on mitochondrial dysfunction.
‘The findings derived from the animal literature feed nicely into my ongoing PhD work, supervised by Professors Siddharthan Chandran and Giles Hardingham, where I am investigating the role of mitochondrial dysfunction, using human induced pluripotent stem cells to study ALS/MND ‘in a dish’. I am very grateful to the MND Association and the MRC for funding this work’ – Dr Arpan Mehta.
Read the full article in Brain Communications.
Read about how our Lady Edith Wolfson Fellowship launched Dr Arpan Mehta’s career in our previous blog post here.
Photo credit: Euan MacDonald Centre





Such exciting research, hopefully the answer to addressing MND even in the latter stages, will be discovered via mitochondrial dysfunction. Congratulations to all those involved.