In June we hosted the second MND EnCouRage UK event for early career researchers (ECR’s) which aims to support them to continue working in the field of MND. The event included lots of talks and workshops from senior researchers to provide tips and advice on moving forward in their careers and help them to develop new skills in areas like presenting their work to non-scientific audiences. We also challenged the ECR’s to write a blog to explain their research to the MND community. This guest blog is from Hannah Bailey, one of the ECR’s who came to the event this year.

I’m Hannah, an MND Association-funded first year PhD student at the University of Nottingham. During my undergraduate degree in Biochemistry, also at Nottingham, I had the opportunity to do a project researching MND in the lab, where my interest in MND began. During this project I also met someone who was living with MND, who had come to hear about the research happening in our lab. Hearing his insights into the devastating effects of MND really stuck with me, and I decided I wanted to begin a career in MND research. This led me to my PhD project, in which I am aiming to make a natural type of cell called astrocytes more protective of motor neurones. We hope that this work will inform us of a possible new way to treat MND.
In the nervous system, astrocytes (named after their star-like shape) are in close contact with motor neurones. Astrocytes are really important for protecting and maintaining the health of motor neurones. Therefore, we can describe healthy astrocytes as ‘neuro-protective’.
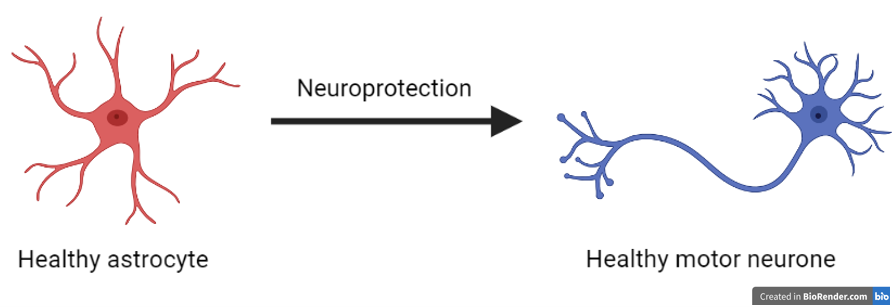
However, in MND, astrocytes can undergo many changes and become what’s called ‘reactive’. These reactive astrocytes often lose the ability to protect motor neurones, instead becoming toxic to them. This makes the neurodegeneration worse.
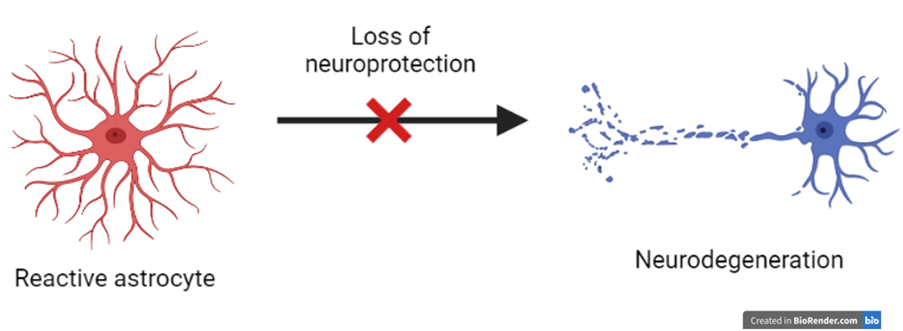
Therefore, a possible future therapeutic strategy that has been considered in MND is to improve the neuro-protective ability of astrocytes. This would allow astrocytes to continue to help protect motor neurones instead of becoming toxic, therefore improving the health of motor neurones.
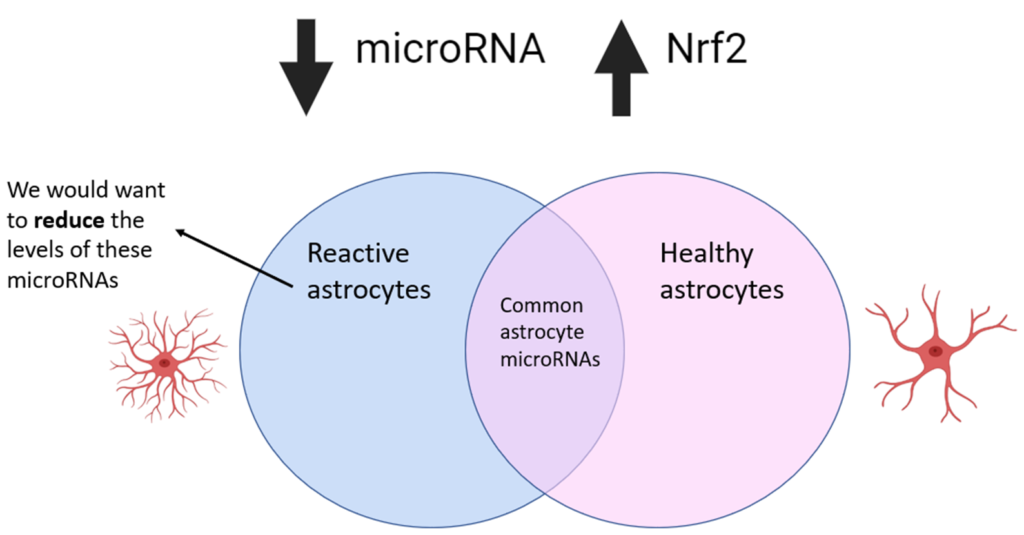
In my work, we are aiming to achieve this by increasing the levels of a protein called Nrf2 in astrocytes. Nrf2’s main job is to protect cells from certain toxic molecules. Importantly, it has been shown that increasing Nrf2 in astrocytes improves their neuro-protective ability.
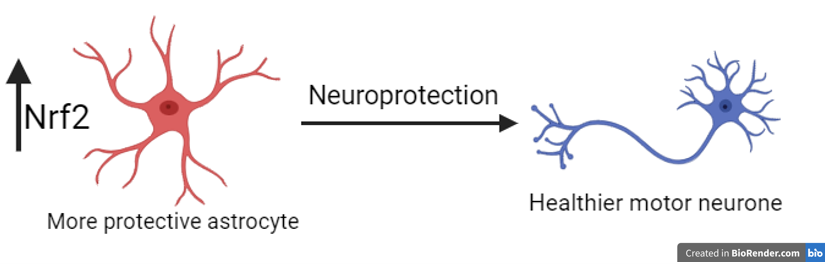
The way that we plan on increasing Nrf2 in astrocytes is by targeting small RNA molecules called microRNAs. microRNAs are found throughout the body, and their job is to change the levels of many proteins. In all cells, we have DNA which contains all our genes. These genes are copied to another type of genetic molecule called RNA, which is then used to make protein. Proteins then do a lot of the important jobs to keep our cells working. The way microRNAs usually work is by preventing RNA from being used to make protein, like the brakes on a car.
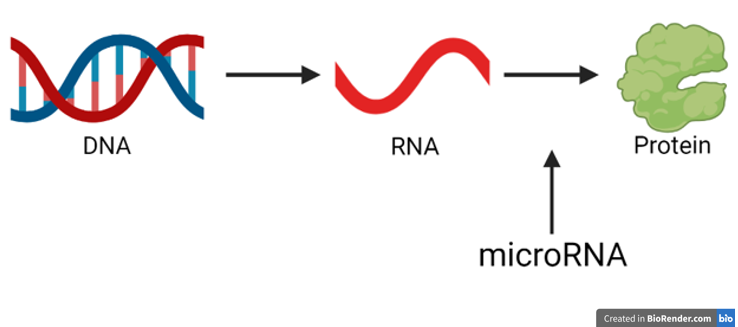
This means that normally, there are certain microRNAs which can prevent Nrf2 protein being made. When we target the right microRNA in astrocytes, this will allow RNA to be used to make Nrf2 protein, and we should see an increase in Nrf2 protein.
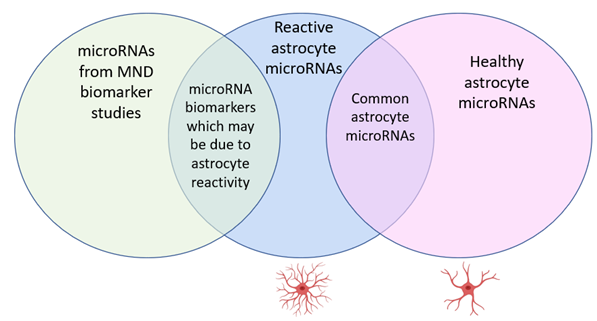
To help us choose the right microRNAs to target in astrocytes, we did an experiment to find out all the microRNAs in human astrocytes – both ‘healthy’ and ‘reactive’ astrocytes. As a general rule, we may want to target microRNAs which are increased in reactive astrocytes compared to healthy astrocytes, as these microRNAs may be causing harm to motor neurones.
As I’ve mentioned, microRNAs are really useful as we can target them to make beneficial changes to proteins. Therapeutics which use RNA are also massively on the rise, the most famous example being the RNA covid vaccine. Therefore, the technology for RNA therapeutics is improving rapidly, supporting the idea that microRNA targeting is a promising strategy for treating MND.
MicroRNAs themselves are also relevant to MND, as the levels of certain microRNAs have been found to be significantly different in people living with MND. microRNAs are really stable in body fluids such as blood and saliva, therefore they could be measured in a relatively non-invasive manner and may be a promising biomarker for diagnosis or to monitor disease progression or response to new treatments.
Part of the work we are doing is to improve our understanding of where these microRNA changes come from in MND. The microRNA data of healthy and reactive astrocytes that we produced is helping us to do this. We can compare this astrocyte microRNA data to other MND clinical biomarker data, to help us understand how astrocytes may contribute to microRNA changes in the body in MND.
To summarise, in my PhD I am working to improve our understanding of astrocyte microRNAs in MND. Using this knowledge, we are targeting specific microRNAs in astrocytes, with the aim of increasing Nrf2 protein. We predict that this will improve the ability of astrocytes to protect motor neurones. If this is the case, then we will show that microRNA targeting in astrocytes may be a promising therapeutic strategy to treat MND, which should be investigated further.
Many thanks to my supervisors Prof Robert Layfield, Dr Daniel Scott, Dr Federico Dajas-Bailador and Dr Sébastien Serres.
We would like to thank Hannah for taking the time to write this guest blog about her work and also thank everyone involved in making MND EnCouRage UK 2023 such a success. Hannah’s PhD project is funded by the MND Association.
Thank you very Hannah!