Global biotechnology company Biogen has announced plans to make tofersen, a potential treatment for people whose MND is believed to be driven by mutations in the SOD1 gene, available to more people on an early access programme. Biogen have also revealed that they are conducting a new Phase 3 clinical trial of tofersen in those who have an SOD1 gene mutation but do not yet show symptoms of MND.
What is tofersen?
People with MND who have a SOD1 gene mutation are thought to produce a faulty SOD1 protein. The protein misfolds in nerve cells (neurons) in the brain and spinal cord and causes the onset of MND symptoms. SOD1 mutations are thought to account for around 2% of all MND, and have been found in both familial and sporadic forms of MND. Tofersen is an antisense oligonucleotide (ASO) which targets the genetic instructions for the faulty SOD1 protein and aims to reduce the amount of this disease-causing protein being produced (read more about how tofersen works in our previous blog).
Tofersen is being developed by global pharmaceutical company Biogen and, following a successful phase1/2 trial (read more in our blog here), is being tested in an ongoing Phase 3 study called VALOR. This phase 3 study is further testing the safety of tofersen in a larger number of participants and is also assessing how effective the treatment is at slowing the progression of the disease. The treatment phase of VALOR, which was being conducted at many different centres worldwide (including Sheffield and London in the UK), has now come to an end with the results still being analysed and to be shared publicly in Autumn this year.
Following completion of the treatment part of the Phase 3 study, an open label extension has been made available for participants to take part in. This means that everyone who completed the Phase 3 trial, including those who received the placebo during the study, have been offered treatment with tofersen for up to five years. This open label extension will help to assess the long-term safety, effectiveness, and tolerability of the treatment.
UPDATE- October 2021:
In a press release published on 17 October 2021, Biogen announced their Phase 3 trial of Tofersen (VALOR) did not meet its primary endpoint.
Despite this, signs of reduced disease progression were seen across multiple secondary and exploratory endpoints, including reduced levels of SOD1 protein in the CSF. Additionally, the results of VALOR and the ongoing open label extension showed that early access to tofersen led to less decline across secondary outcome measures.
From these results, Biogen have announced it will expand its ongoing early access programme to include more people with SOD1-MND who meet the eligibility criteria. This will occur in countries where early access programmes are permitted by local regulations and future access to Tofersen may be secured. This includes the UK, and Biogen is in contact with licensing and regulatory authorities in this country. However, this early access may have to revised or discontinued if another controlled trial is required by regulators or if no path forward can be established for Tofersen.
Biogen are currently working with regulators, the medical community, patient advocacy groups and key stakeholders around the world to to decide what the next steps for Tofersen will be.
Thank you to those with MND who took part in the trial around the world. Our fight for a cure for MND continues.
Early access programme: what does it mean?
Biogen announced in a press release that there are plans to make tofersen available on two different early access programmes to those with SOD1-MND.
The Individual Compassionate Use Access programme enables those with the most rapidly progressing forms of SOD1-MND to gain access to treatment with tofersen from this month. This access has now been granted after all VALOR study participants are no longer randomised to receive the placebo and have transitioned to tofersen on the open label extension, but before the data from the Phase 3 trial has been fully analysed. For those who wish to use this programme, disease progression will be measured by a clinician using the ALSFRS-R and only those with a significant decline in function (a decrease of two or more scale points per month) will be eligible to enrol. Requests to join this access programme must be submitted by a clinician.
The second Early Access Programme for the broad SOD1-MND population is set to begin later this year, after the data from the Phase 3 study has been assessed. If results are positive, whereby tofersen is a safe and effective treatment of SOD1-MND and no further clinical studies are required, Biogen plans to offer tofersen to the wider SOD1-MND community. This access will be offered before applications are submitted to regulatory bodies (such as the Food and Drug Administration and the European Medicines Agency) who assess the safety of new drugs and provide approval for the treatment’s use in clinical practice. The programme will open in countries where it is permitted by local regulations and where Biogen can ensure future access to the treatment for those with SOD1-MND.
The SOD1 gene mutation is estimated to cause around 2% of all MND cases and these programmes could mean that some people with this genetic form of MND will be able to access tofersen treatment this year, before the completion of the formal clinical trials process.
Pre-symptomatic Phase 3 trial (ATLAS)
As well as announcing plans for both early access programmes, Biogen revealed that they are conducting a new Phase 3 study for tofersen, called ATLAS. The ATLAS study seeks to determine the value and optimal timing to begin treatment with tofersen in those who have SOD1 gene mutations but who do not yet have clinical manifestations of MND and are therefore considered pre-symptomatic. This refers to when someone displays underlying signs of the disease, such as nervous system damage (motor neuron damage) but has no symptoms or apparent clinical signs of the disease.
This trial aims to enrol around 150 participants who are known to have one of the
pre-defined list of SOD1 mutations that typically cause MND to rapidly progress. It is planned to be conducted across 30 sites worldwide (UK sites to be confirmed) to see if treatment with tofersen can delay the onset of symptoms, as well as slow the progression of the disease once symptoms appear.
The ATLAS trial consists of four parts (as shown in the picture below) and all participants will start in part A. See the drop-down boxes to read more about each part of the trial:
NfL is a biomarker of nerve cell damage (read more about NfL here) as it leaks out when neurons are dying and is a good indicator of early motor neuron damage that may lead to the development of MND. Levels of NfL have been found to increase before clinical symptoms appear and are a good indicator of pre-symptomatic MND. Previous research looking at those with an SOD1 gene mutation has shown that some people go on to develop MND and this has helped to identify a threshold level for NfL for pre-symptomatic MND for this study. In part A of the study, participants will have monthly blood tests to measure NfL levels and if an increase is found that raises the level to or beyond the threshold level, indicating pre-symptomatic MND, then that individual will move to part B.
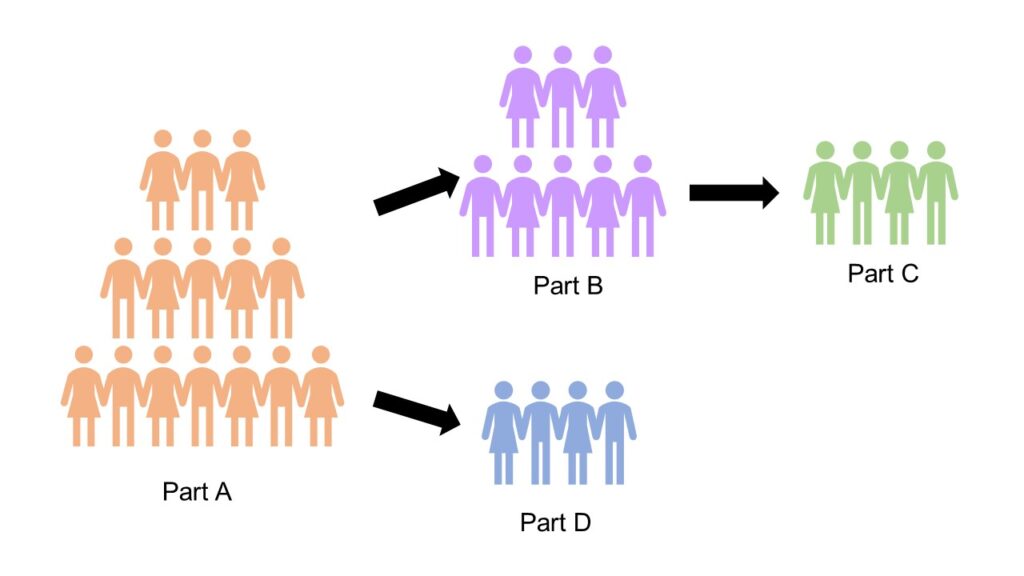
This trial will help to assess the effectiveness of tofersen in delaying the onset of symptomatic MND. The primary endpoint of the study will measure the number of participants that go on to develop clinical symptoms of MND within a year of beginning treatment with tofersen compared to those who are given the placebo. Secondary endpoints of the trial include the percentage of participants who develop clinical symptoms within two years of starting treatment, changes in ALSFRS-R scores and changes in NfL levels throughout treatment.
What does this all mean?
These announcements from Biogen are promising and suggest that tofersen may be an effective treatment for those with SOD1-MND.
“I am cautiously optimistic as pre-clinical work has suggested that this treatment has great potential. It may just be the first effective treatment for some genetic forms of MND and, if successful in the long term, the ability to prevent onset of symptoms before MND ‘starts’ in people will be fantastic – not only for the people and families involved but as a proof of principle and providing a path forward for other potential genetic treatments for the remaining cases with a known genetic cause.”
Nick Cole, Head of Research at MND Association
This summer is an important time for the global pharmaceutical company, with recruitment for the ATLAS trial already underway in some countries (Japan and the U.S.), and the open label extension and individual compassionate use programmes also beginning this month. The data from the Phase 3 VALOR trial is still being analysed and results will be shared on our blog once the findings have been published later on this year.
We are in touch with Biogen and are working with the company to understand what these early access programmes and new trial could mean for our community. An update to this blog will be provided when further information on this is established.
Resources:
- Clinical Trials Website:
- Phase 3 trial VALOR (NCT02623699)
- Open label extension (NCT03070119)
- Presymptomatic SOD1-MND phase 3 trial ATLAS (NCT04856982)
- MND Research Blog:
- Biogen Website:
