I’m Hannah, a PhD student from the University of Nottingham, and a Communications Ambassador for the 34th International Symposium on ALS/MND that took place in December 2023. In this blog post I will be sharing the key messages of a talk at the symposium by Dr Sandra Banack. Dr Banack is a Senior Scientist at Brain Chemistry Labs in Jackson, Wyoming. Her talk was titled “A Robust MicroRNA ALS Blood Diagnostic Test”, and was presented in the Tissue Biomarkers session.
Dr Banack’s research into a new diagnostic test for ALS is vital, as there are currently many issues with diagnosing ALS:
- Diagnosis often takes too long – around 12 months on average
- People can be misdiagnosed
- Diagnosis can be expensive in countries where healthcare is not free
These factors, along with many others, make the wait for an ALS diagnosis an incredibly stressful time for people living with ALS and their families. An effective diagnostic test could help to increase the speed and accuracy of diagnosis. This may allow people living with ALS to receive treatments or participate in clinical trials at an earlier stage, which could have a real positive impact on quality of life. A diagnostic test would also rule out ALS for people who don’t have it.
Dr Banack said that the test must be:
[tabs][tab title=”Blood-based”] This would make the test easy to use and cause minimal discomfort for people getting the test.[/tab][tab title=”Unique to ALS”] The test needs to be able to tell ALS apart from other ‘mimic’ diseases that have similar symptoms.[/tab][tab title=”Reproducible”]The test must always work and give an accurate result, no matter who is taking the blood or running the test.[/tab][/tabs]The aim of the research was to find biomarkers in the blood that could help to diagnose ALS. Biomarkers are naturally occurring molecules in our body that can be measured and tell us whether someone likely has a certain disease. Rather than looking at the blood as a whole, they decided to focus on small packages found in the blood called extracellular vesicles (or EVs). EVs are released from cells all around the body.
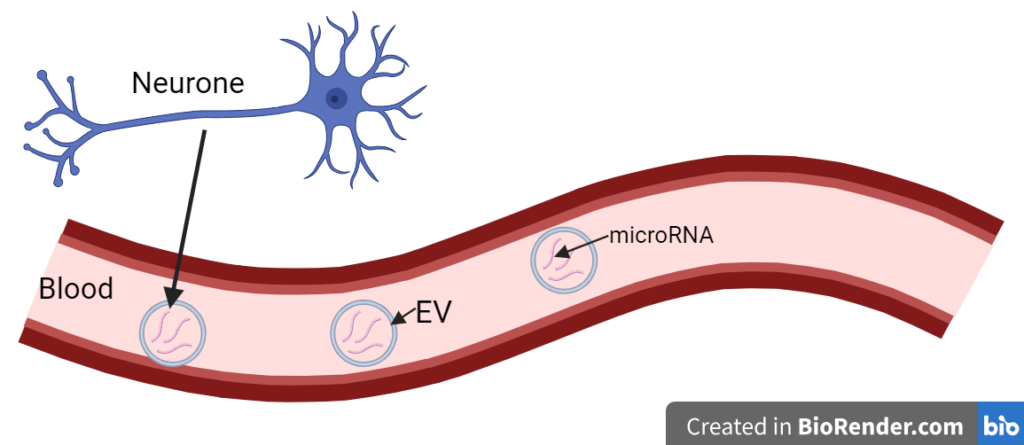
There are several features of EVs that make them a good place to look for biomarkers:
- They are common in body fluids, including the blood.
- Molecules on its surface can give us an information on where in the body the EV came from.
- There are a range of molecules that can be packaged into EVs, such as proteins, lipids (fats) and genetic molecules. Importantly, these molecules are often selected to go into the EV in a non-random way, and disease can influence what is packed into an EV.
- The outer layer of the EV protects its contents so they aren’t destroyed in the blood.
Since neurones are the main cell type affected in ALS, Dr Banack and colleagues decided to look at EVs that have mainly come from neurones. To do this, they selected EVs from blood that had a molecule on their surface that tells us they come from neurones. While this method isn’t perfect at choosing only neurone EVs, it means a larger proportion are from neurones than would otherwise be the case.
As I’ve said, something that can be found in these small packages in the blood are genetic molecules. Dr Banack and colleagues decided to look at a type of very small genetic molecule called microRNAs. Diseases can impact which microRNAs are packaged into EVs. Therefore they decided to compare the microRNAs from EVs of healthy individuals to those from people living with ALS, to see if there were any differences.
Initially lots of microRNAs were found to be at different levels in the healthy EVs compared to ALS EVs. They narrowed this list down to 8 microRNAs – some of which are increased in ALS EVs, some are decreased.
Their first study used 10 blood samples from both healthy participants and people with ALS. The blood was taken in exactly the same way for each person. However it is important that this 8 microRNA ‘fingerprint’ can be found in a larger group of people with ALS, including when the blood samples are taken and prepared in different ways. Therefore repeats of the study were necessary – there have now been 4 repeats, all of which show the same 8 microRNA ‘fingerprint’ in EVs from the blood of people with ALS, even if the blood is taken in different ways. The total number of blood samples studied is now 189 from people with ALS and 220 from healthy people.
Another important consideration is whether the test can tell the difference between people living with ALS and similar neurological conditions. It is unlikely that someone who is healthy would be recommended by a neurologist to be tested for ALS, so it is quite likely that they may have a different neurological condition if not ALS.
Therefore in the most recent study they included blood samples from 42 people with primary lateral sclerosis (PLS), and 20 with Parkinson’s disease. They found that 5 out of the 8 microRNAs were different in Parkinson’s disease compared to ALS. In PLS samples (the disease which is most similar to ALS) all 8 of the microRNAs are different. This is good news as it seems the microRNA test can tell ALS apart from these similar diseases. However Dr Banack emphasised that they need more samples from other similar diseases to confirm that this is true.
The next steps of the research may include:
- Using the test on blood samples from more people with ‘mimic’ (similar) diseases to confirm that the test is specific to ALS.
- Working with diagnostic companies with the aim of making the test available for healthcare professionals to use in the clinic.
- Investigating whether the 8 microRNAs change as the disease progresses.
In summary, Dr Banack and colleagues looked at small packages in the blood called extracellular vesicles, that have mostly come from neurones. Inside these packages there is a type of genetic molecule called microRNA. They found that by measuring the levels of 8 of these microRNAs, we can tell the difference between someone who has ALS and someone who does not.
While this test wouldn’t ever be used on its own to diagnose ALS, it may be used in the future to help confirm a diagnosis if a neurologist thinks that someone may have ALS. Therefore hopefully, this test may help increase the speed and accuracy of ALS diagnosis, which could allow earlier treatment and reduce anxiety while waiting for a diagnosis for people living with ALS and their families.
We would like to thank Hannah for taking the time to write this blog and also for being a Symposium Communications Ambassador. You can follow Hannah on Twitter/X here and read more about Hannah’s research in a previous blog here.
