MND is a complex disease, and the causes are still not fully understood. Researchers all around the world are working tirelessly on different theories to try to understand more about what causes MND and how to treat it. One theory is that MND is caused by the activation of ancient viruses which are hidden within our DNA. To test this theory, a clinical trial investigating the use of retrovirals in people living with MND is currently underway in the UK, Europe and Australia. We recently sat down with Professor Julian Gold to chat about this theory and how HIV drugs could be used to treat MND. Watch more below or read on to find out more.
Introducing Professor Julian Gold…
Professor Julian Gold is an infectious disease specialist, including in the treatment of HIV. He became interested in repurposing HIV drugs for other diseases. Along this journey, he came across research which suggested that MND could potentially be caused by a virus. Professor Gold is the lead investigator of the Lighthouse II clinical trial, which is investigating a HIV drug, called Triumeq, in people living with MND.
Does this mean MND is caused by a virus?
Throughout evolution, humans have picked up ancient viruses which become embedded within our DNA. These viruses, known as retroviruses, infect cells, create copies of their DNA, and then insert this DNA into the hosts DNA. It has been shown 9% of our DNA is made of ancient retroviruses picked up during evolution. Many are defective or lying dormant and historically the DNA from them was considered to be “junk DNA”. However, over the years some of these retroviruses have been linked to diseases. Probably the most well-known retrovirus is the Human Immunodeficiency Virus (HIV), which causes Acquired Immunodeficiency Syndrome (AIDS).
A particular retrovirus, known as human endogenous retrovirus-K (HERV-K) is thought to be activated in the brain and nerve cells of people with MND. Researchers have also shown that mice which are genetically modified to express HERV-K develop MND-like symptoms.
How could HERV-K cause MND?
Researchers are still working on understanding how HERV-K might contribute to the development of MND. A number of different pathways are thought to be involved, but work is still ongoing to understand these pathways and how they are linked to HERV-K.
For example, the activation of HERV-K may be controlled by a protein called TDP-43, which is known to be implicated in MND. When researchers increased the production of TDP-43 in human neurons (grown in a dish), they observed an increase in production of HERV-K. HERV-K has also been shown to increase TDP-43 accumulation, which is observed in 97% of all MND cases. Work is ongoing to understand more about the role of HERV-K and TDP-43 in MND.
Professor Gold explains more about how HERV-K is thought to be involved in MND in a video below.
Could a HIV drug be used to treat MND?
Many people with HIV have also developed neurological problems, including MND-like symptoms. Several studies have documented improvement in these symptoms with anti-retroviral therapy. Hear Professor Gold talk about this more below.
Triumeq is made up of a combination of anti-retroviral drugs. These drugs target different mechanisms of the retrovirus, all with the same goal of preventing the virus from copying itself and implanting into the hosts DNA. When HIV treatments were being developed, they found that targeting more than one mechanism was much more effective than just using one drug.
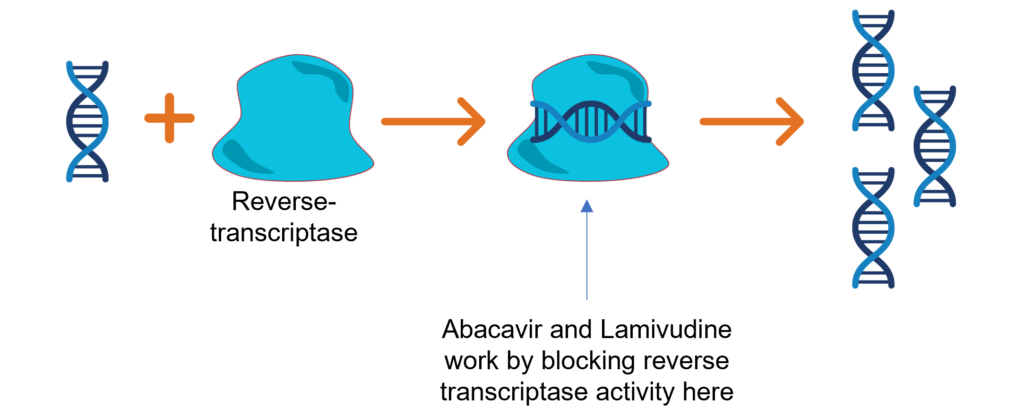
Two of the drugs (Abacavir and Lamivudine) block the activity of an enzyme called reverse-transcriptase. If this enzyme doesn’t function the retrovirus can’t make more copies of itself and the amount of activated retrovirus in the body is reduced.
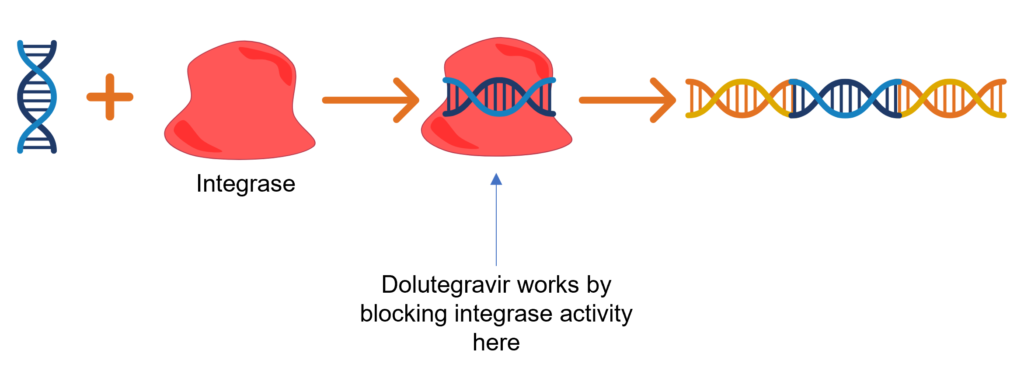
The other drug (Dolutegravir) which makes up Triumeq blocks another enzyme called integrase. This enzyme plays an important role in helping place the viral DNA copy into the hosts DNA. When it is blocked, the viral DNA can no longer be inserted into the human DNA and prevents the retrovirus from replicating.
Has Triumeq been tested in MND clinical trials before?
Triumeq was tested in a phase 2 clinical trial, called Lighthouse. This trial was held in Australia and recruited 40 people with MND who all received a daily dose of Triumeq for 24 weeks. There was no placebo group in this study, so all participants received the treatment. Participants were initially observed for 10 weeks to determine their predicted rate of disease progression. This was then compared with their actual progression whilst on the treatment. Participants were assessed using measures such as the ALS Functional Rating Scale (ALSFRS-R) and breathing capacity (Forced Vital Capacity; FVC). The trial also measured the levels of HERV-K throughout the study, to see if taking Triumeq reduced the amount of virus present.
Generally, the drug was found to be well-tolerated by the participants and safe to administer at the dose it was given. This was to be expected, since Triumeq has already been proven to be safe for the treatment of HIV. There was a statistically significant reduction in disease progression, as measured by ALSFRS-R. Participants were shown to have a change in decline from 1.12 points per month during the initial observation period to 0.76 points per month whilst on the treatment. Breathing progression was also shown to stabilise after 18 weeks on the treatment, indicating another positive change.
In most participants on the trial, levels of HERV-K were reduced, suggesting that the treatment is working as it should within the body. However, it took around 24-weeks for levels to significantly reduce. As this trial was only 24-weeks long, the full potential clinical benefits of Triumeq may not have been observed. This trial provided proof-of-concept of using Triumeq as a potential treatment for MND and lead to the development of a larger, and longer, phase 3 trial (Lighthouse 2).
What is the Phase 3 clinical trial?
Lighthouse II is a phase 3 clinical trial investigating Triumeq in people living with MND. It is a randomised, double-blind, placebo-controlled study, meaning that participants are randomly allocated to receive either the treatment or a placebo without knowing which one they are taking. The trial will assess whether Triumeq is effective in delaying the progression of MND, if it is safe and well tolerated and how it affects health outcomes in people with MND.
What will happen in the Phase 3 trial?
This is a much larger trial than the Phase 2 one and is expected to enrol 390 participants who will take Triumeq or the placebo for a maximum of 24 months. Measurements of disease progression and reaction to the drug will be recorded every 3 months.
Throughout the 24-months there will be 2 interim analyses. This is where data collected from the trial will be independently reviewed, by people not involved in the trial. This provides opportunities to stop the trial early if the drug is not showing positive signs for those living with MND. We know that people living with MND don’t have time to wait, so trials that include interim analyses are vital for making sure participants spend the minimum amount of time on trials while also thoroughly testing new potential treatments.
How can I take part in the trial?
The trial has been designed to allow more people living with MND an opportunity to take part in a clinical trial. For example, there is no time limit since onset of symptoms to take part in this trial. Potential participants will need to have a blood test to check that they are not likely to have an allergic reaction to one of the anti-retrovirals.
The trial is aiming to recruit up to 200 participants in the UK. Recruitment is currently taking place at:
- King’s College London
- University College London
- Sheffield
Additional centres are currently in set-up around the country. For more information on recruiting sites use the button below to go to our website or contact us on research@mndassociation.org.
As Professor Gold mentioned in our interview, Dr Avindra Nath has played a vital role in this research. Dr Nath and team have delved deeper into how a virus might cause MND in the laboratory, increasing knowledge of this area and helping provide evidence for the clinical trials. You can find out about Dr Nath’s work in a webinar from FightMND.
We would like to thank Professor Gold for contributing to this blog.
King’s College London is the lead trial site in the UK. The MND Association is proud to support to this trial site by funding a research nurse.