A recent press release by the pharmaceutical company Biogen reported preliminary results from an ongoing clinical trial investigating a form of precision therapy in people with SOD1-related MND. This drug, known as tofersen, is now in the final stages of Phase 1/2 testing in centres across the world, including Sheffield in the UK.
Tofersen is an antisense oligonucleotide (ASO), designed to prevent the faulty disease-causing protein from being made. Proteins, the building blocks of the body, are created from our genetic information (DNA) via its photocopy (RNA). If a piece of DNA is damaged, the RNA will also be damaged, leading to formation of a faulty protein and creating issues in the body. Tofersen is a synthetically-created RNA directed to stick to the faulty photocopy (RNA) preventing it from making faulty proteins.
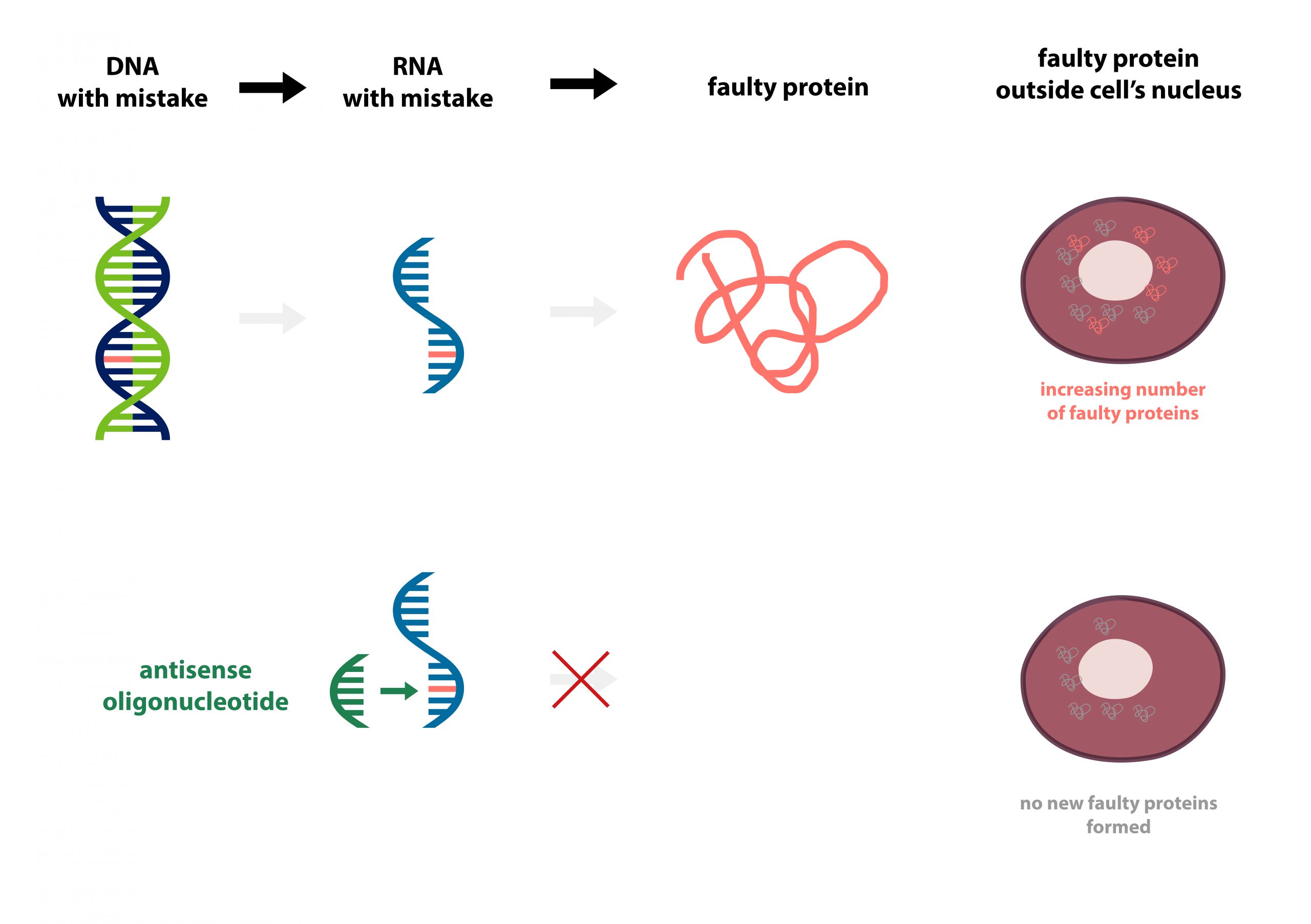
As the study is in its early stages, it is mainly focused on the safety and dose of the drug. However, the inclusion of a double blind design, a placebo arm and a relatively high number of participants (38 who received tofersen and 12 placebo) has helped to provide an insight into the drug’s functionality.
The study has shown that people receiving the highest dose of the drug (100mg) during the three month trial had significantly reduced amounts of SOD1 protein, compared to those taking the placebo, and signs that the progression of their MND had slowed down – measured by the ALSFRS-R disease progression rating scale, slow vital capacity and muscle strength.
A Phase 3 trial is already recruiting participants in the USA. This will explore the effects in a greater number of participants. In a separate clinical trial, a similar drug is also being investigated for its safety and, eventually, effectiveness, in people with C9ORF72-related MND – this trial plans to recruit people in the UK in the future.

Dr Nick Cole, Head of Research at the MND Association, commented: “The data is encouraging and if the final outcomes are as positive as we hope, this would be a significant milestone in the fight against MND. This therapy would be the first targeted treatment for a specific type of genetic MND, bringing hope to people and families with one type of inherited MND.
“This study demonstrates that through continuing public support, collaboration and partnerships between charities, researchers and pharmaceutical companies we will find solutions to the difficult question of MND.
“Although this treatment may only be suitable for a relatively small proportion of people with MND, any positive outcome resulting in a therapy is hugely significant and would mean so much to a lot of people.
“The hope is that this type of targeted technology could be applied to other genetic forms and, ultimately, pave the way to uncovering treatments for sporadic MND, which represents the vast majority of cases.”







Please talk in layman terms and add “inherited type mind” at the start of your posts. Do not buidpoples hopes only to be dashed at he bottom of the article!!
Dear Martin,
Our aim is to inform the public about important findings in the field of MND research in an accurate manner, and it definitely isn’t our intention to build up hopes and then dash them. I appreciate that some articles might feel as if they are distant from finding a cure for MND, however, all of these findings are bringing us closer to finding THE treatment, step by step.
As we mentioned at the end of the article, it is also important to remember that even though some drugs target specific genes, it doesn’t mean they are irrelevant to most people with MND, as these techniques might be key to treating all forms of the disease.
Best Wishes,
The Research Development Team
Tofersen I am on this treatment at Sheffield Hallamshire Hospital and I believe I am doing extremely well on it I have the drug monthly via a lumber puncture.All the staff are brilliant and I can’t prraise them highly enough