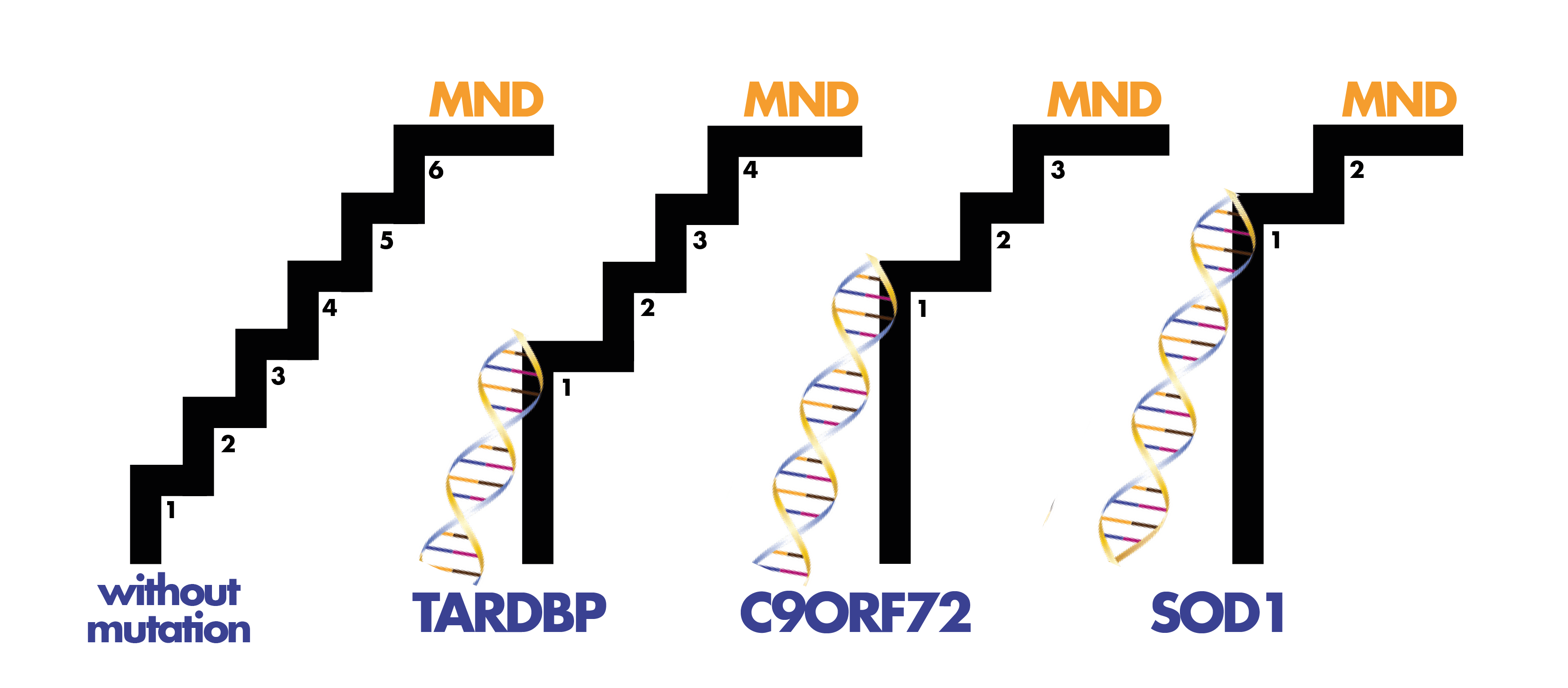
Genetics and MND
Reading Time: 5 minutes This is blog number two in our ‘Symposium Blogathon’ – counting down to the 32nd International Symposium on ALS/MND. Numbers in bold green text correspond to the code in the abstract book. Click on the number to be redirected to the full abstract (the page may take a minute to load). Approximately 10% of people…