Building relationships with key research institutes across the UK is vital in pushing research forward in the fight against MND. As of 31 December 2023, we are funding 109 research projects across 19 UK Universities and research institutes.
The MND Association has a long-standing relationship with University College London (UCL) and its researchers. This relationship has helped forge new connections, including with The Francis Crick Institute in London, a flagship centre for discovery research in biomedicine. These institutes work closely together at the forefront of MND research to increase our understanding of what’s going wrong in the body in the disease and investigate new potential treatments. Currently, we are funding 14 research projects across these institutions.
The team recently visited The Francis Crick Institute to hear the latest research updates, meet our MND Association funded researchers and tour the labs where the pioneering research happens. In this blog we give an overview of some of the exciting research we heard about.
Understanding what goes wrong in MND
Before we can find effective treatments of MND, we first need to understand the biology of the disease to know what it is that is going wrong. Researchers have uncovered many different key pathways involved in MND. However, as MND is so complex, there is still much more to learn about the disease. A significant amount of the research happening at UCL and The Francis Crick Institute focuses on furthering our understanding of the disease, which could potentially lead to the development of new treatments. Read on to learn more about some of these projects.
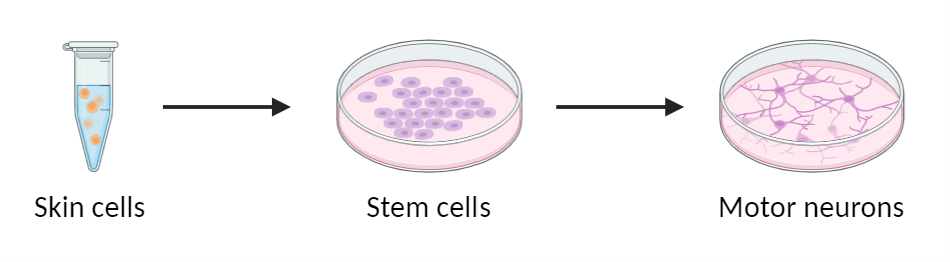
Using motor neurons ‘grown in a dish’
As MND affects the brain and spinal cord, it is impossible to take samples from people living with the disease without causing damage. Instead, researchers can now grow cells, such as motor neurons in a Petri dish. This can help them understand what goes wrong in MND and where. Lady Edith Wolfson Clinical Fellow, Professor Rickie Patani and team are using this induced pluripotent stem cell (iPSC) technology to ‘trick’ skin cells from people with MND to become stem cells. These stem cells and then be transformed into any cell within our bodies, including motor neurons. The team are also looking to understand how ageing makes motor neurons vulnerable to MND, as MND is more common in people above the age of 50.
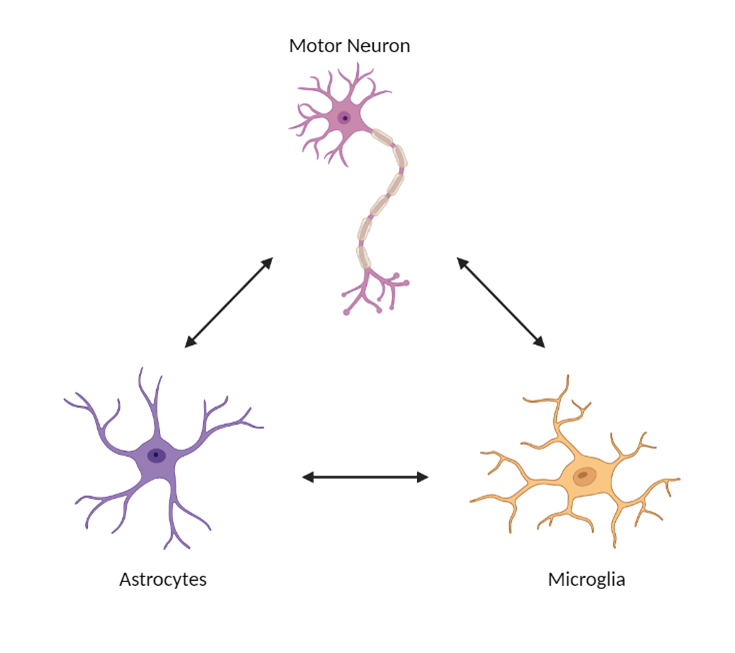
How do cells surrounding and supporting motor neurons contribute to motor neuron death in MND?
Previous research has suggested that cells which support and surround motor neurons, known as microglia and astrocytes, can contribute to motor neuron death. Dr Ben Clarke is investigating whether the cells work together incorrectly to cause damage. Dr Clarke is using the same method as Professor Patani to grow cells ‘in a dish’. He will then look for changes that happen in MND compared to people without the disease. Once any changes have been identified, he will try to see if the changes can be corrected. Dr Clarke will also investigate the interactions between astrocytes, microglia and motor neurons and see if one cell type helps to drive the disease in the others.
Can motor neuron damage be caused by disruptions in how proteins are made?
Proteins are vital in performing all the key tasks needed to keep a cell healthy. When proteins are made differently, this can stop them from performing tasks correctly and can lead to cell death. One key protein is FUS, which plays a crucial role in controlling how other proteins are made. FUS is known to be faulty in some people with MND and this disrupts the production of other proteins. Dr Nicol Birsa is investigating how this disruption alters the function of motor neurons and how they connect to muscles. Once it is understood how motor neurons are damaged, Dr Birsa will investigate if it is possible to correct the damage.
RELATED POST
Blog | 14 February 2022 | Charlotte Roy
Silence is golden: A new clinical trial for FUS-ALS
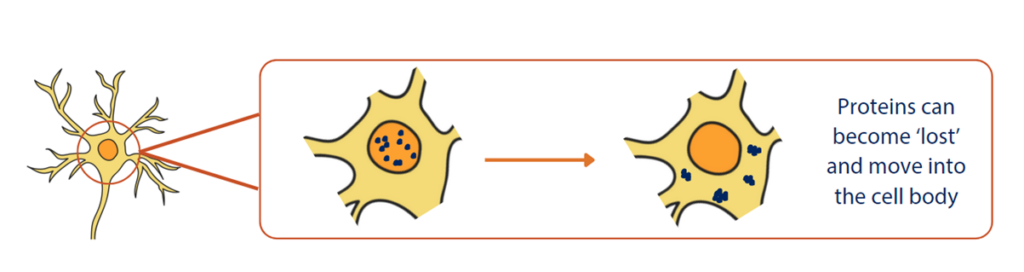
How do ‘lost’ proteins contribute to MND?
In MND, proteins can become ‘lost’ and move from the control centre of the cell (called the nucleus) to the cell body (called the cytoplasm). This movement means the proteins are no longer able to function as they should. This loss of function can corrupt the formation of other proteins.
Proteins are made from DNA, which acts as the instruction manual. Using these instructions, DNA creates messages, known as RNA. Parts of these messages (called introns) must be removed to make healthy proteins. This is like removing the adverts from a movie so that you can watch it properly. The ‘adverts’ are not needed for the complete functional story of the movie but do disrupt and impact the overall quality of the finished product. Research has shown that in MND, the interfering RNA messages ‘introns’ are not removed, which could contribute to the development of the disease. Dr Yiran Wang aims to learn more about the pathways that lead to proteins becoming ‘lost’ and why introns are not removed in MND.
Developing potential new treatments
Once researchers have identified pathways which go wrong in MND, they can then start working on how to correct them or prevent damage from happening. This is the first step in the long process of developing potential new treatments. Lady Edith Wolfson Clinical Fellow, Professor Pietro Fratta, and his team are using their recent findings to begin to develop a new cutting-edge gene therapy.
Research has shown that a protein called TDP-43 is in the wrong place in nerve cells of 97% of people with MND. Therefore, developing a therapy which targets TDP-43, to prevent this misplacement from happening, could be beneficial for many people with MND. The teams previous research has shown that faulty TDP-43 can cause mistakes in the instructions which make other proteins. As a result these proteins can also become faulty.
One of these proteins, called UNC13A, plays an important role in how motor neurons communicate. When UNC13A doesn’t work as it should it is thought this could contribute to motor neuron damage. The researchers are testing different small DNA-like molecules to see how effective they are at correcting the mistakes in the instructions to make UNC13A. The most promising gene therapy could then be further developed and potentially tested in clinical trials.
Find out more about the research we fund
This is just a small snapshot of the innovative research that the MND Association is funding. We have relationships with key research institutes all across the UK and, through the organisation of the International Symposium on ALS/MND, we have relationships with researchers all across the world. It is through these relationships that we can continue to fund the most promising and cutting-edge research, which will help us achieve our goal of a world free of MND. To find out more about the research we fund head over to our website.
