A newly published paper in Acta Neuropathologica Communications has shed light on the behaviour of healthy motor neurones compared to motor neurones found in MND. This work looks to provide another crucial piece to the puzzle of what goes wrong with motor neurones in MND. The movement of substances along the motor neurones is known as axonal transport and it is thought that this important transport system is one of the elements that goes wrong in the affected motor neurones in MND.
The lead author of the research was one of our MND Association funded Lady Edith Wolfson Fellows, Dr Andrew Tosolini, from University College London. Below you can hear from Andrew as he answers some questions about this research.
What did this work focus on?
Motor neurones are a type of nerve cell that enable us to move by transmitting signals from our brain and spinal cord to the muscles in our body. These are the main type of cells affected by MND. In our central nervous system, which is made up of the brain and spinal cord, there are different types of motor neurones. There are fast neurones that activate muscles for performing precise movements, such as those in our hands, and there are slow motor neurones that control muscles that maintain posture, such as those in our back and abdomen. For reasons still to be fully understood, fast motor neurones are more vulnerable to MND, whereas slow motor neurons remain relatively unaffected. One theory for fast motor neurones being more vulnerable to MND is that axonal transport is selectively disrupted and this research investigated this theory using a model of MND. Before I explain more about axonal transport, let’s first look at what an axon is.
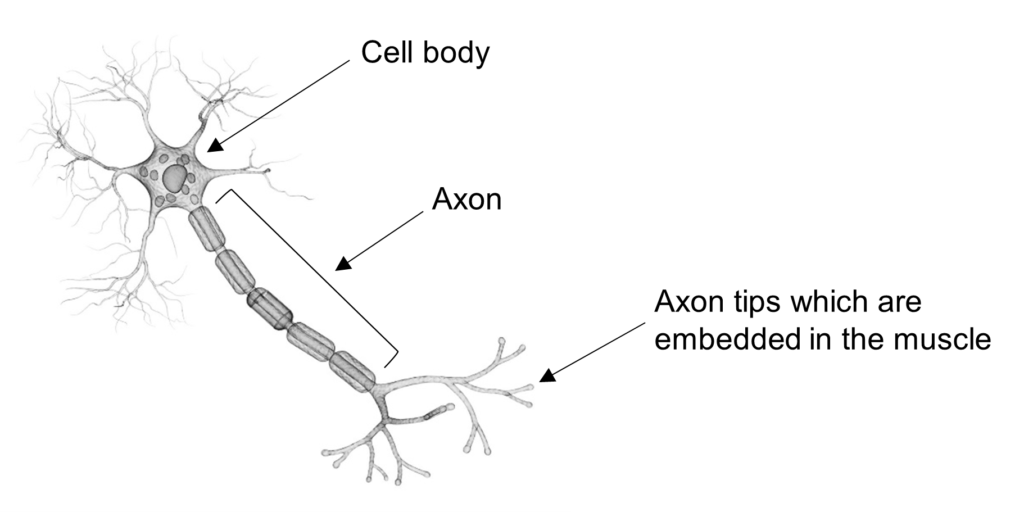
What is the axon?
The command centre of a motor neurone, known as the cell body, sits within our spinal cord. To be able to activate muscles, motor neurones have a long, thin extension that arises from the cell body and projects out to connect to the muscle, this is known as the axon. The motor neurone uses the axon to send electrical signals to the muscle to trigger muscle contractions.

Interesting Fact
Axons in motor neurones can be up to a metre in length and a fraction of a milimeter in diameter, making them too thin to be seen with the naked eye.
What is axonal transport, and why is it important?
Electrical impulses are not the only signals that are transmitted along axons. Neurones use a process called axonal transport to move many important cellular substances up and down the axon (figure 1a). Axonal transport is essential for the nerve to survive and function properly.
In one direction, the mitochondria, which are like batteries and provide energy to the nerve cells, need to be moved from the cell body towards the muscle. In the opposite direction, critical survival factors are transported along axons to the cell body where they help to keep motor neurones healthy. The video below shows axonal transport in a mouse motor neurone.
Disruptions in axonal transport are a common feature of many neurodegenerative diseases, including MND and dementia, suggesting that defects in this process contribute to the degeneration of neurones.
What did you find in this research?
We found that in healthy mice, axonal transport is similar in both fast and slow motor neurons. However, when axon terminals were stimulated with a protein called brain-derived neurotrophic factor (BDNF), an important survival factor for motor neurones, axonal transport speeds increased in fast, but not slow, motor neurones (figure 1b).

Interesting Fact
The word ‘neurotrophic’ is derived from a combination of two ancient Greek words and literally means ‘nerve nourishing’
We then assessed axonal transport in a mouse model of MND called SOD1G93A, and found that axonal transport is disrupted, but only in the fast motor neurones (figure 1c). Intriguingly, when we stimulated the fast motor neurones of the MND mouse model with BDNF, it didn’t increase the speed of axonal transport (figure 1d).
These results highlight a difference between axonal transport in healthy motor neurones and motor neurones in MND. They suggest that the crucial BDNF survival signalling pathway is impaired specifically in vulnerable fast motor neurons in MND.
We also showed that in MND, there is an imbalance in the number of key proteins that recognise and bind to BDNF (i.e., receptors) in both muscles and nerves. This provides a possible explanation for the loss of response to BDNF.
Figure 1
What does this mean for people with MND?
The exact cause of motor neuron death in MND is still unclear. The more we understand about what is going wrong, the better we can develop targeted therapies to slow and prevent MND. Using this important mouse model, we have discovered a new key deficit in MND, hence this work has added another crucial piece to the puzzle of what goes wrong in neurones in MND.
What do you think are the next steps for this work?
The next critical steps will be to determine whether axonal transport deficits are common to the fast motor neurones in other mouse models of MND, as well as in human motor neurones grown in a dish. If it can be confirmed that slow motor neurones are not affected in all types of MND, this would suggest that slow motor neurones are somehow resistant to the disease or that something is making fast motor neurones more vulnerable to damage. By comparing the properties of slow and fast motor neurones, we may be able to find explanations for their differences in MND and could investigate ways to specifically treat the vulnerable population of fast motor neurones.
RELATED TOPIC
Blog | 10 December 2019 | Mandy Spencer
Highlighting MND researchers of the future
How has your MND Association fellowship helped you?
The MND Association continues to play a crucial role in facilitating progress in the fight against MND. In 2020, I was awarded the Lady Edith Wolfson Junior Non-Clinical Fellowship, which has been instrumental to my work dedicated to understanding how axonal transport contributes to MND. I am extremely grateful to the MND Association for its support, because without it, I would not have been able to follow this important line of research, and our discoveries would not have been made available to the research community. Moreover, the MND Association has provided additional vital funding to Professor Schiavo’s laboratory (where I work), along with other members/laboratories of the Department of Neuromuscular Disorders at the Queen Square Institute of Neurology, University College London.
We wish to thank Andrew for taking the time to talk to us about his research.
The MND Association would like to thank the Batchworth Trust, the Payne-Gallwey Charitable Trust, the Edwin George Robinson Charitable Trust, the Inman Charity and the Sylvia Aitken Charitable Trust for their generous support of this project.
Additional resources:
Dr James Sleigh, who worked on this project, has written a short (and free to read!) article on axonal transport for non-scientists which you can find here.